
REHOVOT, ISRAEL — March 10, 2026 — RNA technology is regarded as one of the newest frontiers in medicine, but in fact a primordial innovator got there way before we did. The malaria parasite, an ancient single-celled organism, has been using sophisticated RNA maneuvers for millennia. In a study recently published in Cell Reports, researchers at the Weizmann Institute of Science uncover the parasite’s RNA strategies – mechanisms that could inspire unexpected applications for RNA-based tools in multiple areas of medicine.
More than a decade ago, Prof. Neta Regev-Rudzki discovered that the malaria parasite Plasmodium falciparum, which invades human red blood cells, sends out tiny vesicles to communicate with fellow parasites inside other red blood cells. These nanovesicles – wrapped, sac-like packages addressed to other cells – were shown to transfer pieces of DNA from one parasite to another. But Regev-Rudzki and her team found that the vesicles also contain other molecular cargo, including various types of RNA.
The researchers reasoned that the clever parasite wouldn’t transport its RNAs via vesicles without good reason. In the new study, the team, in Weizmann’s Biomolecular Sciences Department, set out to determine whether the vesicles enable the RNAs to perform tasks beyond messaging. Because the team had previously shown that nanovesicles enter not only red blood cells but also immune cells called monocytes, the researchers suspected the parasite might be trying to mess with the host’s immune defenses.
They soon learned that the parasite is even more devious than they had imagined. The team, led by Dr. Paula Abou Karam, then a PhD student, found that the RNAs in the vesicles were not random fragments but rather messenger RNA, or mRNA – specifically, molecules containing instructions for producing one of the parasite’s most abundant protein families. Could the parasites be inserting their own mRNA into the cells’ protein-making machinery in the cytoplasm?
Contrary to expectations, there was no sign of the parasitic proteins they expected these RNAs to make in the cytoplasm of the monocytes. Instead, to the researchers’ surprise, the RNAs appeared to be entering the immune cells’ highly protected nuclei.
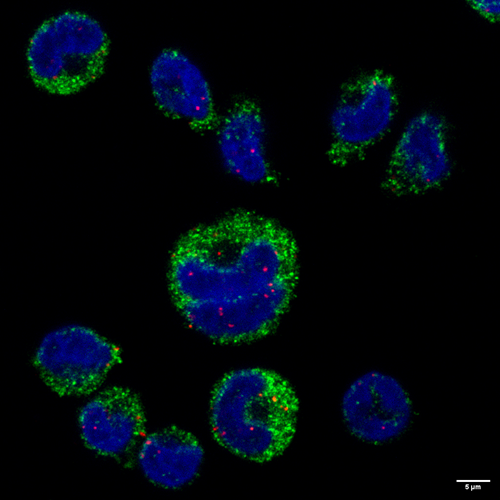
“This seemed incredible,” Regev-Rudzki recalls. “The cell guards its nucleus jealously because it houses the cell’s ‘brain.’ To convince ourselves – and others – that the parasite’s RNAs truly penetrate these defenses, we had to observe them directly inside the nucleus.”
At first, this task seemed nearly impossible. Parasitic RNA molecules are present in extremely small amounts that are easily lost within the crowded nucleus. Another difficulty stemmed from the parasite’s genome itself, which is unusually repetitive, composed largely of just two genetic letters, A and T. Designing a method sensitive enough to detect the parasite’s RNAs amid all the host genetic material was thus a huge challenge.
“Sometimes what makes or breaks a research project is the student who leads it, and this was one of those classic cases,” Regev-Rudzki says. “Paula took on the challenge, keeping at it for a year and a half until she ultimately succeeded.”
Abou Karam developed a fluorescent probe that caused minuscule red dots to light up inside the monocyte nucleus. Each dot represented a single malarial mRNA molecule that had breached the nuclear barrier.
The next question was: What were these RNAs doing there? The researchers discovered that once inside the nucleus, the parasite mRNA binds to two human proteins, ACIN1 and PNN – key components of the cellular splicing machinery. Like splicing film, cells use the splicing to edit RNA transcripts, cutting and rearranging segments before allowing them to be translated into proteins. It serves as a crucial quality-control step: Without proper splicing, RNAs do not “make sense,” and they are generally discarded before they can be translated into proteins.
“The study’s implications extend far beyond infectious disease."
At this stage, the researchers joined forces with the team of Dr. Zeev Melamed of the Hebrew University of Jerusalem, an expert in splicing and RNA. Together, they revealed splicing manipulation in recipient monocytes, thereby deciphering the parasite’s RNA strategy.
Sabotage in the nucleus
In effect, the parasite jams the internal communications of the host’s immune system. By getting its mRNA into monocyte nuclei, it disrupts the host cell’s own RNA processing. The result is chaos. Critical immune transcripts – meant to produce proteins that fight infection – are improperly spliced and then sent off to be degraded. As a result, the production of entire families of immune proteins is shut down.
As they continued to observe the immune cell takeover, the scientists found that the manipulated monocytes send out distress signals, triggering a wave of immune activation that mobilizes additional immune cells. But while these cells rush to deal with the apparent crisis inside the monocytes, the real threat – parasites quietly multiplying inside red blood cells – escapes attention.
“It’s a decoy mechanism,” Regev-Rudzki says. “Like throwing a grenade in one direction so the guards run toward it, while you move somewhere else.” By confusing immune cells and suppressing key defense proteins, the parasite buys itself precious time to grow and spread.
These findings point to a potential new target for antimalarial drugs – therapies designed to prevent malarial RNAs from tampering with the host’s splicing machinery.
The work may also open new avenues for diagnosis, not only of malaria but of other infectious diseases. Bilharzia parasites, for example, can reside in the gut for years, causing damage while remaining undetectable in standard blood tests. Regev-Rudzki believes that they, and other parasites, may be releasing vesicles whose RNA signatures in the bloodstream might reveal their presence.
The study’s implications extend far beyond infectious disease. Vesicles abound in the body and searching for their cargo not only in the cytoplasm, but in unexpected places such as the carefully guarded nuclei, can increase the chances of intercepting these packages and reading their messenger RNA. And that, in turn, might help us decipher disease-promoting mechanisms. The feints and strategies employed by cancer, for example, include the release of vesicles that influence surrounding healthy tissue. Vesicles are also an active area of research in Parkinson’s, ALS, and other neurodegenerative diseases. Vesicle-bound RNAs circulating in blood may one day serve as early biomarkers for these disorders.
Also participating in the study were Edo Kiper, Shaked Yadid, Dr. Ewa Kozela, Nir Zharoni, Dr. Reinat Nevo, Daniel Alfandari, Helina Otesh, Abel Cruz Camacho, and Dr. Ofer Shoshani of Weizmann’s Biomolecular Sciences Department; Dr. Tamar Ziv of the Technion – Israel Institute of Technology; Dr. Yoav Lubelsky and Prof. Igor Ulitsky of Weizmann’s Immunology and Regenerative Biology Department; Drs. Ron Rotkopf, Ekaterina Petrovich-Kopitman, and Ziv Porat of Weizmann’s Life Sciences Core Facilities Department; Dr. Eviatar Weizman of the Nancy and Stephen Grand Israel National Center for Personalized Medicine; Moshe Cossin of the Hebrew University of Jerusalem; Dr. Irit Rosenhek-Goldian of Weizmann’s Chemical Research Support Department; and Prof. Carmit Levy of Tel Aviv University.
Prof. Neta Regev-Rudzki’s research is supported by the Henry Chanoch Krenter Institute for Biomedical Imaging and Genomics; the Dr. Barry Sherman Institute for Medicinal Chemistry; the Kleinman Cancer Cell Sorting Facility; the Henri Gutwirth Award; the Karen Siem Fellowship for Women in Science; and the Brenden-Mann Women’s Innovation Impact Fund
